Antagonist Raw Material Ondansetron
Basic Information
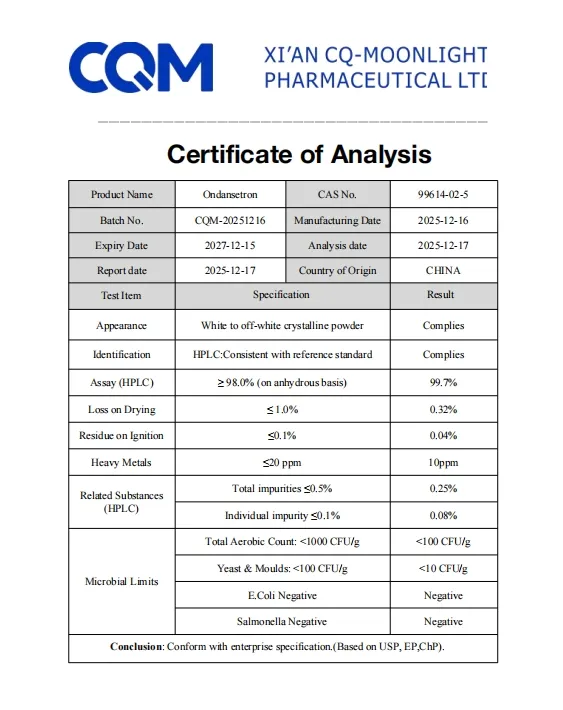
| Model NO. | CQM-Ondansetron |
| Certification | GMP, HSE, ISO 9001, USP, BP |
| Suitable for | Elderly, Children, Adult |
| State | Powder |
| Purity | >99% |
| CAS | 99614-02-5 |
| Molecular Formula | C18H19N3O |
| Appearance | White Powder |
| Assay | 99% |
| Test Method | HPLC |
Product Description
Ondansetron is a highly selective 5-hydroxytryptamine 3 receptor antagonist that can effectively block the release of 5-hydroxytryptamine induced by chemotherapeutic drugs, radiotherapy or surgical stimulation, thereby inhibiting central and peripheral emetic reflexes. It is widely used in the prevention and treatment of nausea and vomiting associated with radiotherapy and chemotherapy as well as postoperative nausea and vomiting.
Application & Function
Ondansetron acts on relevant receptors in the gastrointestinal tract and the central nervous system. By blocking signal transduction, it inhibits the excitation of the afferent pathway of the vagus nerve. Unlike some traditional antiemetic drugs, it has fewer adverse effects on the cardiovascular system and the central nervous system. It can be administered through multiple routes including oral administration and intravenous injection, making it a first-line choice for antiemetic treatment.
Specification & COA
Product Display
Frequently Asked Questions
What is the purity of your Ondansetron powder?
Our Ondansetron powder maintains a high purity level of over 99%, tested via HPLC to ensure pharmaceutical grade quality.
What certifications does the product have?
The product is compliant with GMP, HSE, ISO 9001, USP, and BP standards, ensuring global regulatory compliance.
What are the primary applications for this material?
It is primarily used as a raw material in the pharmaceutical industry to treat and prevent nausea and vomiting induced by chemotherapy, radiotherapy, and surgery.
How is the product transported and packaged?
The transport package is negotiable to meet specific client requirements, ensuring the stability and safety of the powder during transit.
Does Ondansetron affect gastrointestinal motility?
No, Ondansetron exerts its antiemetic effects without affecting normal gastrointestinal motility or the conscious state of the patient.
What is the production capacity for this product?
We have a stable production capacity of approximately 1000kg per month to meet large-scale pharmaceutical needs.

























 Health Line
Health Line