














| Still deciding? Get samples of $ ! US$ 800/kg Request Sample |

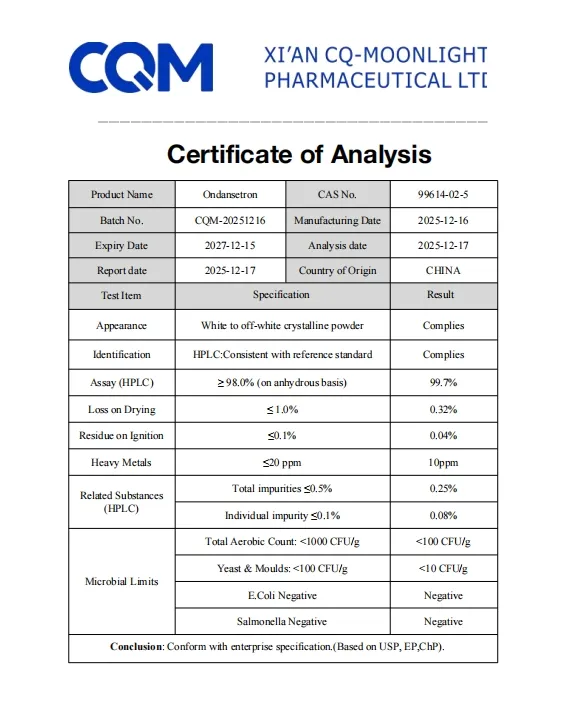
| Model NO. | CQM-Ondansetron | Certification | GMP, HSE, ISO 9001, USP |
| Purity | >99% | State | Powder |
| CAS No. | 99614-02-5 | Molecular Formula | C18H19N3O |
| Appearance | White Powder | Melting Point | 231 - 232ºC |
| Application | Pharmaceutical Grade | Production Capacity | 1000kg/Month |
Ondansetron is a highly selective 5-hydroxytryptamine 3 receptor antagonist that can effectively block the release of 5-hydroxytryptamine induced by chemotherapeutic drugs, radiotherapy or surgical stimulation, thereby inhibiting central and peripheral emetic reflexes. It is widely used in the prevention and treatment of nausea and vomiting associated with radiotherapy and chemotherapy as well as postoperative nausea and vomiting.

Ondansetron mainly acts on relevant receptors in the gastrointestinal tract and the central nervous system. By blocking signal transduction, it inhibits the excitation of the afferent pathway of the vagus nerve, effectively alleviating nausea and vomiting reactions. Unlike traditional antiemetic drugs, it has fewer adverse effects on the cardiovascular and central nervous systems. It can be administered via oral or intravenous routes, making it a reliable first-line choice for clinical antiemetic treatment.





 Health Line
Health Line